Infectious Coryza in poultry: A mini-review

Dr. Uddab Poudel
Department of Veterinary Microbiology and Parasitology, Agriculture and Forestry University (AFU), Rampur, 44209, Nepal
Abstract:
Infectious coryza, which is one of the major problematic respiratory disease of poultry is caused by a well differentiated gram negative, potentially pathogenic bacterium; Avibacterium paragallinarum, and characterized by nasal discharge, sneezing, and facial swelling with the complex clinical sign of pneumonia and air-sacculitis in secondary infections. Disease has worldwide distribution and particularly affect laying birds by decreasing egg production up to 40% and declined growth performance in broilers as well. Bacterial culture or Polymerase chain reaction (PCR) is used for diagnosis. All-in/all-out of flocks, sound management practices and use of vaccines can help in prophylaxis. Immediate administration of antibiotics particularly erythromycin and oxytetracycline can reduce severity and course of infection.
Key words: antibiotics, facial swelling, infectious coryza, nasal discharge
Introduction:
Infectious Coryza (IC) or snot is an acute to chronic infectious upper respiratory tract bacterial disease of the poultry characterized by decreased activity, nasal discharge, sneezing, and facial swelling with fetid odor of exudates (Pat J. Blackall & Soriano-Vargas, 2019; García, Angulo, Blackall, & Ortiz, 2004; Hobb et al., 2002). The disease was infectious and primarily affects nasal passage so, it was named as infectious coryza. The disease primarily occurs in chickens as they are natural host for the disease; but disease is occasionally seen in indigenous native domestic fowls, pheasant, guinea fowls, Japanese quails, numida and parrots (CABI, 2019; Cigoy, Huberman, & Terzolo, 2016; Priya, Krishna, Dineshkumar, & Mini, 2012; Wahyuni, Tabbu, Artanto, Setiawan, & Rajaguguk, 2018).Chicken of all age group are highly susceptible and mature birds are at more risk and occurrence of disease is higher in chickens farming in intensive system (Charlton, 2000). The disease is often seen during the flock’s peak egg laying season and when birds are in stress condition (Williams & Fulton, 2019). Outbreaks are more frequent during fall and winter seasons (Soriano-Vargas, 2021; Vargas & Terzolo, 2004).
Etiology
Etiological agent responsible for the occurrence of disease is Avibacterium paragallinarum; formerly called Haemophilus paragallinarum which is classified under genus Avibacterium (Patrick J. Blackall, Christensen, Beckenham, Blackall, & Bisgaard, 2005; Cigoy et al., 2016; Mei et al., 2020).The genus Avibacterium contains other four different species of bacteria where only Avibacterium paragallinarum is considered to be pathogenic and Avibacterium endocarditis and Avibacterium gallinarum are opportunistic pathogen and remaining two Avibacterium avium and Avibacterium volantium are non-pathogenic (Pat J. Blackall & Soriano-Vargas, 2019; Patrick J. Blackall et al., 2005). As early as 1920, Beach believed that infectious coryza was a distinct entity. The etiological agent eluded identification for a number of years because the disease appeared in mixed infection particularly with fowl pox. In 1932, De Blieck isolated the causative agent and named it Bacillus hemoglobinophilus coryzae gallinarum. However, in 1934, Elliot and Lewis, and Delaplane independently proposed the binomial Haemophilus gallinarum as it required X - (haemin) and V- (nicotinamide adenine dinucleotide, NAD) factors for growth. In 1962, Page found that all the isolates recovered from the cases of infectious coryza required only V- Factor for growth and proposed new species Haemophilus paragallinarum. Moreover, studying genotypic and phenotypic investigation of the taxonomy of Haemophilus paragallinarum, it was reclassified as Avibacterium paragallinarum (Patrick J. Blackall et al., 2005; CABI, 2019).
Avibacterium paragallinarum is a gram- negative, pleomorphic, non-motile, microaerophilic bacteria that requires NAD (nicotinamide adenine dinucleotide) (V-factor) for in vitro culture and organisms are able to reduce nitrates and utilize carbohydrates (figure-1) (Ali, Hossain, Akter, Khan, & Hossain, 2013; Charlton, 2000; Soriano-Vargas, 2021). Bacteria grow in blood agar but require nurse colony of Staphylococcus aureus that excretes v-factor (Zuku-review, 2021). However, few isolates discovered in South Africa, Peru and Mexico are NAD independent and highlight, the need for biochemical test or new generation of PCR for identification (CABI, 2019; Soriano-Vargas, 2021).
Figure-1: The causative bacterium of infectious coryza is Avibacterium paragallinarum, a gram-negative, pleomorphic, non-motile and microaerophilic rod
Photo obtained from: Ali, M., Hossain, M., Akter, S., Khan, M., & Hossain, M. (2013). Pathogenesis of Infectious Coryza in Chickens (Gallus gallus) by Avibacterium paragallinarum Isolate of Bangladesh. The Agriculturists, 11(1), 45.
Avibacterium paragallinarum is classified under two systems, the Page and Kume systems. According to the Page scheme, Avibacterium paragallinarum is classified into 3 serogroups termed as A, B and C which is based on plate agglutination method. However, Kume method is based on hemagglutination inhibition test and recognized nine serovars: A-1, A-2, A-3, A-4, B-1, C-1, C-2, C-3 and C-4 (Pat J. Blackall & Soriano-Vargas, 2019; Cigoy et al., 2016). Regarding the pathogenicity, few studies suggest A and C are pathogenic and B are non-pathogenic serovars of the bacteria (Dereja & Hailemichael, 2017).
Economics and Public Health significance:
Infectious coryza causes massive economic losses due to poor growth in young birds (due to diarrhea, poor consumption of feed and water), increased number of culls due to dermatitis and cellulitis, and a significant (10 to 40 percent) drop in egg production leading to financial liability to chicken farmers (P. J. Blackall, 1999; Williams & Fulton, 2019). Avibacterium paragallinarum has no public health significance as disease is non-zoonotic. No evidence of human cases have been reported till date (Pat J. Blackall & Soriano-Vargas, 2019).
Epidemiology:
Infectious coryza has economic significance in different parts of the world and disease occurrence is worldwide (Table-1) (Rajurkar, Roy, & Yadav, 2009). Potential impact of coryza in broilers meat has been encountered after an outbreak in California and southeastern states of the United states where unusual clinical signs have been reported (Droual, Bickford, Charlton, Cooper, & Channing, 1990). Outbreak of infectious coryza in a broilers farm of Alabama caused 69.8% condemnation of birds due to severe air sacculitis (Hoerr, Putnam, Rowe-Rossmanith, Cowart, & Martin, 1994). In a study of 10 coryza outbreaks in Morocco, it was found that there was a drop in egg production up to 40% and mortality ranges from 0.7-10%. In an epidemiological study about cause of death of native chickens in Thai village found that 6.8 % of birds under 2 months, 0.7% birds of 2-6 months and 16.8% birds over 6 months died due to infectious coryza (Thitisak, Janviriyasopak, Morris, Srihakim, & van Kruedener, 1988). In 1984, disease was first recognized in China and disease mostly affected Beijing, Guan Zhou, Tianjin, Shandong, Shanghai, Sichuan and Yunnan Province with morbidity ranges from 20-50% and mortality from 5-20%. Within a period of three years (1986-1988), disease caused heavy economic losses of around 100 million yuan in China (approximately $18 million at the 1992 exchange rate) (Chen, Zhang, Blackall, & Feng, 1993). Incidence and outbreak of infectious coryza is less problematic in developed countries compared to developing countries.
Table-1: Distribution table for positive cases of Infectious Coryza in different parts of world
|
Continent/ Region |
Countries |
References |
|
Africa |
Angola, Botswana, Cabo Verde, Cameroon, Egypt, Eritrea, Ethiopia, Ghana, Guinea, Morocco, Seychelles, South Africa, Tanzania, Zimbabwe |
(Ali et al., 2013; Byarugaba et al., 2007; CABI, 2019; Chen et al., 1993; Crispo et al., 2019; Dereja & Hailemichael, 2017; Droual et al., 1990; García et al., 2004; Hobb et al., 2002; Mendoza-espinoza et al., 2009; OIE-Handistatus, 2005; Poernomo, Sutarma, Rafiee, & Blackall, 2000; Priya et al., 2012; Rajurkar et al., 2009; Terzolo et al., 1993; Wahyuni et al., 2018; Xu et al., 2019) |
|
Asia |
Bahrain, Bangladesh, China, Hongkong, India, Indonesia, Iraq, Jordan, Malaysia, Myanmar, Philippines, South Korea, Sri Lanka, Taiwan, Thailand, Vietnam |
|
|
Europe |
Germany, Ireland, United Kingdom |
|
|
North America |
Canada, Cuba, Dominica, Jamaica, Guatemala, Mexico, Panama, United States, Saint Vincent and the Grenadines |
|
|
South America |
Argentina, Bolivia, Brazil, Chile, Colombia, Ecuador, Paraguay, Peru, Uruguay |
|
|
Oceania |
Australia, French Polynesia, New Caledonia, New Zealand |
Transmission
The birds which are chronically ill and healthy carrier birds are main reservoir of infection of Avibacterium paragallinarum. Though all age group are susceptible, susceptibility increases with age.
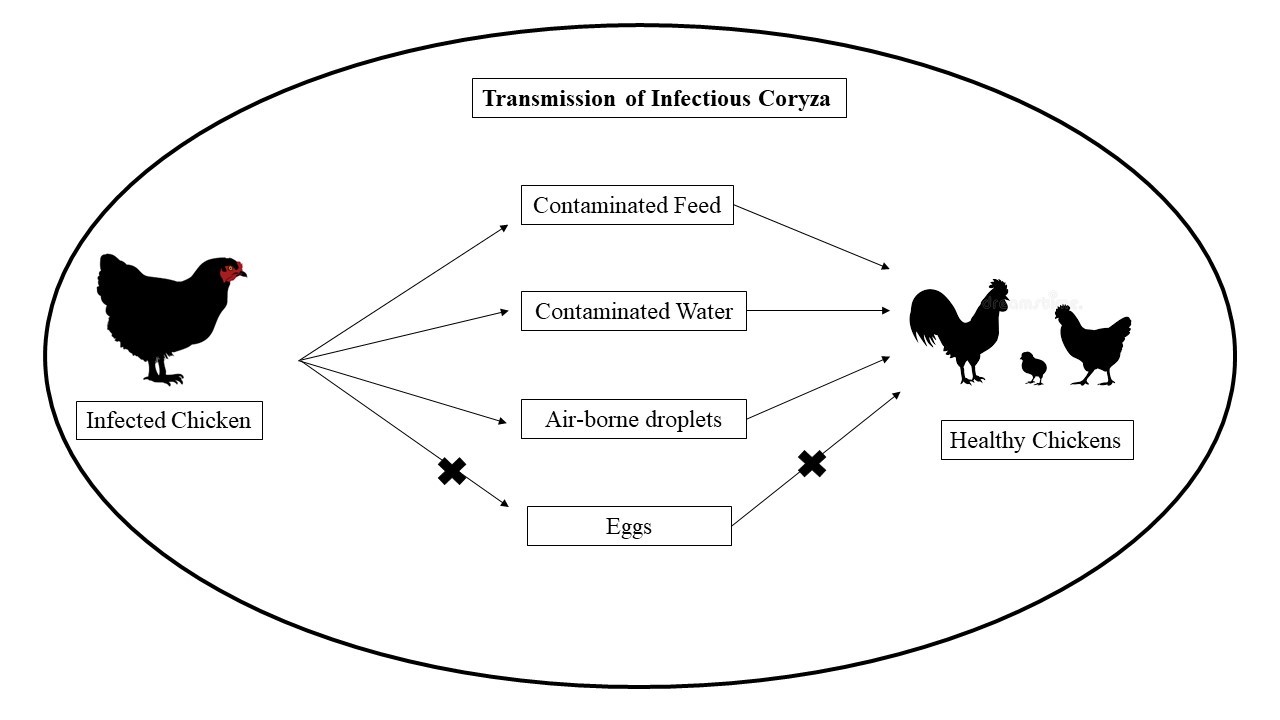
Figure-2: Transmission of Infectious Coryza from infected flock to healthy flocks
Infected chickens create constant threat to healthy flocks. After recovery also, the birds continually harbor Avibacterium paragallinarum without showing any clinical signs. Disease is transmitted by direct contact with contaminated feed, drinking water and air-borne droplets (figure-2). Only horizontal transmission but no evidence of vertical transmission has been found. Therefore, all in – all out management practices help to break transmission chains and disinfection of all the materials before adding new next flocks help to control transmission. That’s why the farms without such flow and with multiple age flocks encounter outbreak of disease more frequently (CABI, 2019; Dereja & Hailemichael, 2017; Soriano-Vargas, 2021).
Clinical Signs
The incubation period (IP) for disease is 1-3 days and typical duration of disease persists for 2-3 weeks. However, the period varies in experimental inoculation of birds with live Avibacterium paragallinarum or the infectious exudates through different routes. Through intranasal inoculation, IP was 24 hour, nasal inoculation 48 hours, 3 days for birds in cage, 4 days for infected drinking water contact and 6-14 days for air- borne transmission of bacteria (Vargas & Terzolo, 2004). Disease has low mortality and high morbidity (Pat J. Blackall & Soriano-Vargas, 2019).
Clinical signs occur in two forms: mild form usually occurs in young chickens and severe form in adults and older birds. In mild form of coryza, signs may be listlessness, depression, a serous nasal discharge and mild facial swelling (Soriano-Vargas, 2021; Zuku-review, 2021). The affected birds reduced feed and water consumptions. In severe form, there will be swelling of the infraorbital sinus with sneezing, serous, mucoid or suppurative nasal discharge. The facial edema prevents eyelids from fully opening leading to conjunctivitis and epiphora. In males, oedematous swelling in intermandibular spaces and wattles is seen. Birds may have varying degrees of rales due to pneumonia and air sacculitis. In some commercial broilers, uncommon signs of otitis and meningoencephalocele were observed (Crispo et al., 2018). There is massive reduction in egg production in layers and delayed egg laying in young pullets. Moreover, presence of secondary pathogens especially, mycoplasmas and virus, and stress factors can result in overall disease complexity (Pat J. Blackall & Soriano-Vargas, 2019). On the basis of clinical signs in head, the infected birds are divided into four grades (Figure-3).
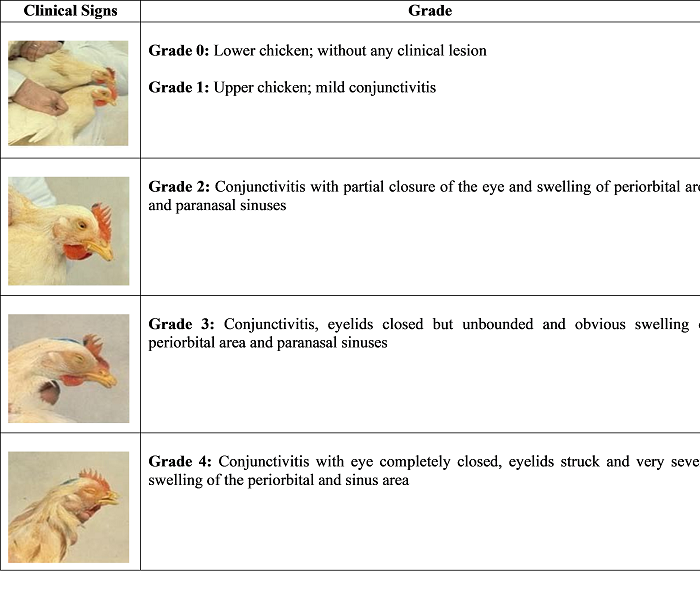
Figure-3: Grading of birds according to clinical signs in head
Photos obtained from: Vargas, E. S., & Terzolo, H. R. (2004). Epizootiology, prevention and control of infectious coryza. Veterinaria Mexico, 35(3), 272.
Postmortem Lesions and Histopathology
In acute cases, the lesions are limited to infraorbital sinuses and nasal passages. Complicated signs may be observed in chronic ones. The lesions may include catarrhal conjunctivitis, congestion of mucous membrane of nasal cavity, yellowish sinus exudates, subcutaneous oedema of face, wattles and intermandibular region. Increase in mucus and necrotic debris in trachea is observed during postmortem (Dereja & Hailemichael, 2017; Soriano-Vargas, 2021). Other lesions may include tracheitis, bronchitis, pneumonia, lung oedema and opacity, and air sacculitis in the case of secondary bacterial or viral infections (Dereja & Hailemichael, 2017). Under histological examination, lesion ranging from necrosis of respiratory epithelium to edema and hyperplasia of sinuses, disintegration and edema of glandular epithelium with infiltration of heterophils, macrophages and mast cells can be found (CABI, 2019; Soriano-Vargas, 2021).
Diagnosis:
Clinical diagnosis of infectious coryza can be done either by bacterial culture or by PCR (Soriano-Vargas, 2021). However, history, clinical signs and symptoms can be suggestive for the diagnosis. Isolation of bacteria is performed from mucus samples taken from the infraorbital sinus of infected birds. Growth of short rods or coccobacilli 1–3 mm in length and 0.4–0.8 mm in width gram negative, satellitic, catalase- negative organism in 24 hours gives confirmatory diagnosis for infectious coryza (Pat J. Blackall & Soriano-Vargas, 2019). Avibacterium paragallinarum shows satellite phenomenon with a feeder culture of Staphylococcus aureus or Staphylococcus epidermidis or Staphylococcus hyicus (CABI, 2019). Though bacterial culture diagnoses the bacteria, PCR has been reported to provide more accurate results. Moreover, inoculation of nasal exudate from infected chicken to susceptible chicken producing typical lesions also helps for diagnosis. Hemagglutination inhibition test exists for serological test but not suitable compared to other diagnostic methods (Soriano-Vargas, 2021).
Prevention and Control:
Disease can be prevented with best management practices following strict biosecurity and vaccination (Figure-4). All recovered birds remained as main carrier of infectious coryza so, eliminating the carriers and depopulating the infected birds, quarantine of new entries plays a pivotal role in prevention of disease. It is recommended to buy chicks and breeding birds from known sources which have no history of infection (CABI, 2019). Frequent turning and changing of poultry litter, proper cleaning and disinfection of the equipment, spraying houses and surrounding premises with suitable disinfectants also helps in prevention and control of the disease. After cleaning and disinfection, it should remain vacant for 2-3 weeks before restocking with new birds (Pat J. Blackall & Soriano-Vargas, 2019; Dereja & Hailemichael, 2017). All-in/ all- out flow of flocks with no multi age group of birds help in prevention of disease (Soriano-Vargas, 2021).
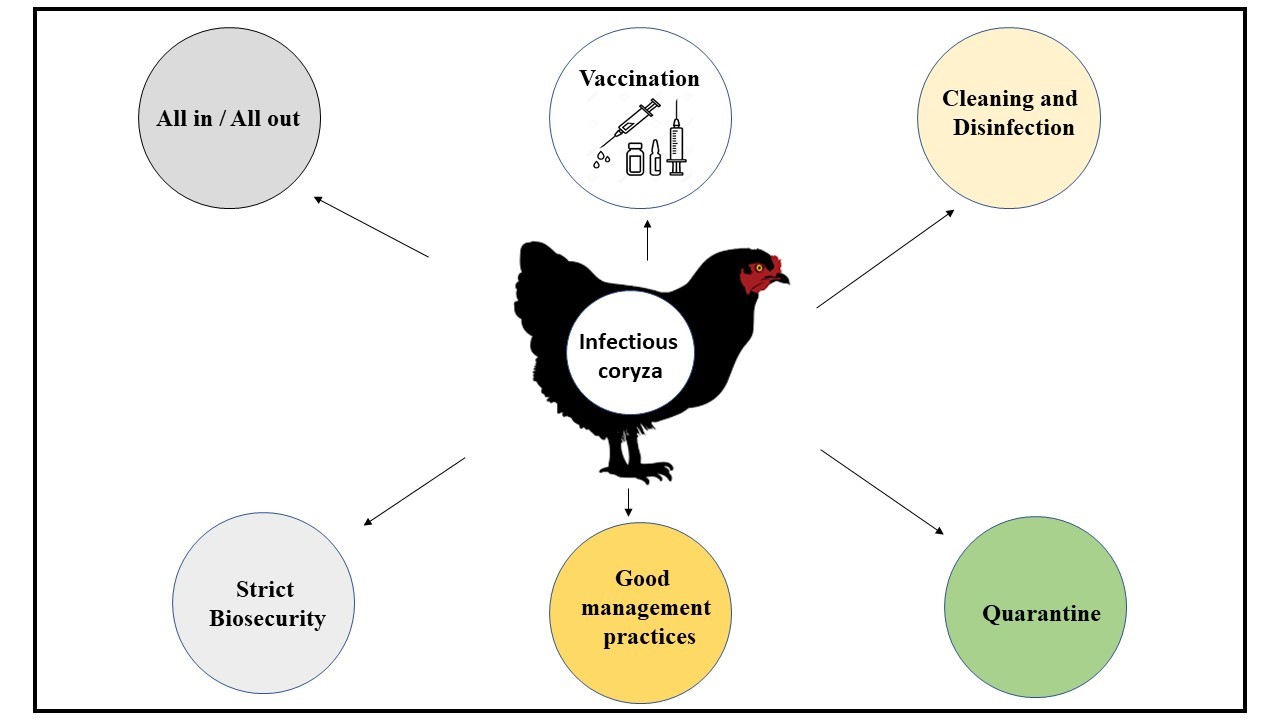
Figure-4: Prevention and control measures for infectious coryza
Bacterins/vaccines are better prophylactic tools (Soriano-Vargas, 2021). Early vaccination usually 4 weeks before the typical outbreak period is recommended. Serovars A, B and C are not cross protective so bacterins should contain serovars present in the target population. All birds on endemic farms should be vaccinated. In some cases, controlled exposure to live organisms also induced protective immunity in layers (Pat J. Blackall & Soriano-Vargas, 2019; Dereja & Hailemichael, 2017; Soriano-Vargas, 2021). Numerous outbreaks have been reported in vaccinated flocks in several countries as well, which suggest further research for novel vaccines and new approaches for vaccine delivery in future.
Treatment:
Early treatment with supportive care is highly important for alleviating the severity and course of infection of infectious coryza. Various chemotherapeutics and antibiotics can be administered through feed or drinking water for successful treatment of infectious coryza (Cigoy et al., 2016). Infected birds respond to treatment however, disease may relapse when treatment is discontinued and the carrier state of infected birds will persist for long time. So, at least for a week enrofloxacin at 10 mg/kg body weight or amoxicillin at 20 mg/kg body weight should be given. Sulfonamides, in combination with other drugs like trimethoprim- sulfamethoxazole or sulphachloropiridazine-trimetroprim via drinking water also give good results (CABI, 2019; Cigoy et al., 2016). Sulfa drugs cause a temporary drop in egg production and overdose may lead to renal toxicity. In case of secondary infection, especially with mycoplasma spp., enrofloxacin (20-25 mg/kg) or kanamycin (30 mg/kg) with gentamicin (5-8 mg/kg) or tylosin tartrate (30 mg/kg) with streptomycin at 100 mg/kg should be given (Cigoy et al., 2016). Use of streptomycin can cause severe stress in the young chicken for about one day. Erythromycin and oxytetracycline are commonly used and effective two antibiotics for treatment of disease (CABI, 2019; Soriano-Vargas, 2021).
References:
Ali, M., Hossain, M., Akter, S., Khan, M., & Hossain, M. (2013). Pathogenesis of Infectious Coryza in Chickens (Gallus gallus) by Avibacterium paragallinarum Isolate of Bangladesh. The Agriculturists, 11(1), 39–46. https://doi.org/10.3329/agric.v11i1.15240
Blackall, P. J. (1999). Infectious coryza: Overview of the disease and new diagnostic options. Clinical Microbiology Reviews, Vol. 12, pp. 627–632. https://doi.org/10.1128/cmr.12.4.627
Blackall, Pat J., & Soriano-Vargas, E. (2019). Infectious coryza and related bacterial infections. In Diseases of Poultry (pp. 890–906). https://doi.org/10.1002/9781119371199.ch20
Blackall, Patrick J., Christensen, H., Beckenham, T., Blackall, L. L., & Bisgaard, M. (2005). Reclassification of Pasteurella gallinarum, [Haemophilus] paragallinarum, Pasteurella avium and Pasteurella volantium as Avibacterium gallinarum gen. nov., comb. nov., Avibacterium paragallinarum comb. nov., Avibacterium avium comb. nov. and Avibacterium . International Journal of Systematic and Evolutionary Microbiology, 55(1), 353–362. https://doi.org/10.1099/ijs.0.63357-0
Byarugaba, D. K., Minga, U. M., Gwakisa, P. S., Katunguka-Rwakishaya, E., Bisgaard, M., & Olsen, J. E. (2007). Virulence characterization of Avibacterium paragallinarum isolates from Uganda. Avian Pathology, 36(1), 35–42. https://doi.org/10.1080/03079450601102947
CABI. (2019). Infectious coryza. Retrieved June 15, 2021, from https://www.cabi.org/isc/datasheet/79285
Charlton, B. R. (2000). Infectious Coryza. In Avian disease manual (5th ed., pp. 94–96). Retrieved from https://www.worldcat.org/title/avian-disease-manual/oclc/52349236
Chen, X., Zhang, P., Blackall, P. J., & Feng, W. (1993). Characterization of Haemophilus paragallinarum Isolates from China. Avian Diseases, 37(2), 574–576.
Cigoy, M. L., Huberman, Y. D., & Terzolo, H. R. (2016). Infectious coryza. Retrieved June 15, 2021, from Engromix, Poultry Industry, Technical Article website: https://en.engormix.com/poultry-industry/articles/infectious-coryza-t39047.htm
Crispo, M., Blackall, P., Khan, A., Shivaprasad, H. L., Clothier, K., Sentíes-Cué, C. G., … Stoute, S. (2019). Characterization of an outbreak of infectious coryza (Avibacterium paragallinarum) in commercial chickens in central California. Avian Diseases, 63(3), 486–494. https://doi.org/10.1637/19-00081.1
Crispo, M., Sentíes-Cué, C. G., Cooper, G. L., Mountainspring, G., Corsiglia, C., Bickford, A. A., & Stoute, S. T. (2018). Otitis and meningoencephalitis associated with infectious coryza (Avibacterium paragallinarum) in commercial broiler chickens. Journal of Veterinary Diagnostic Investigation, 30(5), 784–788. https://doi.org/10.1177/1040638718792964
Dereja, I. A., & Hailemichael, D. (2017). Infectious Coryza in Jimma Backyard Chicken Farms: Clinical and Bacteriological Investigation. Journal of Veterinary Science & Technology, 08(01). https://doi.org/10.4172/2157-7579.1000412
Droual, R., Bickford, A. A., Charlton, B. R., Cooper, G. L., & Channing, S. E. (1990). Infectious coryza in meat chickens in the San Joaquin Valley of California. Avian Diseases, 34(4), 1009–1016. https://doi.org/10.2307/1591398
García, A. J., Angulo, E., Blackall, P. J., & Ortiz, A. M. (2004). The presence of nicotinamide adenine dinucleotide-independent haemophilus paragallinarum in México. Avian Diseases, 48(2), 425–429. https://doi.org/10.1637/7104
Hobb, R. I., Tseng, H. J., Downes, J. E., Terry, T. D., Blackall, P. J., Takagi, M., & Jennings, M. P. (2002). Molecular analysis of a haemagglutinin of Haemophilus paragallinarum. Microbiology, 148(7), 2171–2179. https://doi.org/10.1099/00221287-148-7-2171
Hoerr, F. J., Putnam, M., Rowe-Rossmanith, S., Cowart, W., & Martin, J. (1994). Case report: infectious coryza in broiler chickens in Alabama. Retrieved June 26, 2021, from https://agris.fao.org/agris-search/search.do?recordID=US9532851
Mei, C., Sun, A. H., Blackall, P. J., Xian, H., Li, S. F., Gong, Y. M., & Wang, H. J. (2020). Component Identification and Functional Analysis of Outer Membrane Vesicles Released by Avibacterium paragallinarum. Frontiers in Microbiology, 11, 518060. https://doi.org/10.3389/fmicb.2020.518060
Mendoza-espinoza, A., Terzolo, H. R., Delgado, R. I., Amparo, I., Koga, Y., Huberman, Y. D., … Huberman, Y. D. (2009). Serotyping of Avibacterium paragallinarum Isolates from Peru. Avian Diseases, 53(3), 462–465.
OIE-Handistatus. (2005). World Animal Health Publication and Handistatus II ( data set before 2005). Retrieved June 23, 2021, from Office International des Epizooties website: https://web.oie.int/hs2/report.asp
Poernomo, S., Sutarma, Rafiee, M., & Blackall, P. J. (2000). Characterisation of isolates of Haemophilus paragallinarum from Indonesia. Australian Veterinary Journal, 78(11), 759–762. https://doi.org/10.1111/j.1751-0813.2000.tb10447.x
Priya, P. M., Krishna, S. V., Dineshkumar, V., & Mini, M. (2012). Isolation and characterization of Avibacterium paragallinarum from ornamental birds in Thrissur , Kerala Isolation and characterization of Avibacterium paragallinarum from ornamental birds in Thrissur , Kerala. International Journal of Life Sciences, 1(3), 87–88. Retrieved from http://www.crdeep.com/category/ijlswww.crdeep.com
Rajurkar, G., Roy, A., & Yadav, M. M. (2009). An overview on Epidemiologic investigations of Infectious coryza. Veterinary World, 2(10), 401–403. Retrieved from www.veterinaryworld.org
Soriano-Vargas, E. (2021). Infectious Coryza in Chickens - Poultry - Veterinary Manual. Retrieved June 20, 2021, from https://www.msdvetmanual.com/poultry/infectious-coryza/infectious-coryza-in-chickens
Terzolo, H. R., Paolicchi, F. A., Sandoval, V. E., Blackall, P. J., Yamaguchi, T., & Iritani, Y. (1993). Characterization of Isolates of Haemophilus paragallinarum from Argentina. Avian Diseases, 37(2), 310–314.
Thitisak, W., Janviriyasopak, O., Morris, R. S., Srihakim, S., & van Kruedener, R. (1988). Causes of death found in an epidemiological study of native chickens in Thai villages. Acta Veterinaria Scandinavica. Supplementum, 84, 200–202.
Vargas, E. S., & Terzolo, H. R. (2004). Epizootiology, prevention and control of infectious coryza. Veterinaria Mexico, 35(3), 261–279.
Wahyuni, A. E. T. H., Tabbu, C. R., Artanto, S., Setiawan, D. C. B., & Rajaguguk, S. I. (2018). Isolation, identification, and serotyping of Avibacterium paragallinarum from quails in Indonesia with typical infectious coryza disease symptoms. Veterinary World, 11(4), 519–524. https://doi.org/10.14202/vetworld.2018.519-524
Williams, Z., & Fulton, M. (2019). Poultry Diseases: Infectious Coryza. Retrieved from https://www.canr.msu.edu/resources/poultry-diseases-infectious-coryza
Xu, Y., Cheng, J., Huang, X., Xu, M., Feng, J., Liu, C., & Zhang, G. (2019). Characterization of emergent Avibacterium paragallinarum strains and the protection conferred by infectious coryza vaccines against them in China. Poultry Science, 98(12), 6463–6471. https://doi.org/10.3382/ps/pez531
Zuku-review. (2021). Zuku Review FlashNotes TM Infectious Coryza. Retrieved from https://zukureview.com/sites/default/files/docs/Zuku_Visual_Flashnotes_Infcoryza_extended.pdf

