INFECTIOUS LARYNGOTRACHEITIS IN POULTRY: AN OVERVIEW

Dr. Srikanth Vallabhaneni,
M.V.Sc. Scholar (Department of Livestock Production Management), College of Veterinary Science, Tirupati,
Sri Venkateshwara Veterinary University, Tirupati, Andhra Pradesh, India- 517502. Corresponding Author: srikanthvety20@gmail.com @+917989487841
ABSTRACT:
Infectious laryngotracheitis (ILT) is an extremely infectious viral upper respiratory illness caused by the Gallid herpesvirus 1 (GaHV-1) of the genus Iltovirus and subfamily Alphaherpesvirinae of the Herpesviridae family. ILTV is disseminated via the ocular and respiratory routes. Conjunctivitis, oculo-nasal discharge, bloody mucus, sinusitis, swollen orbital sinuses, respiratory distress, high morbidity, substantial mortality, and diminished egg production are all the clinical manifestations of the disease. To detect ILTV samples from naturally infected or experimentally infected birds, advanced biotechnological technologies such as Polymerase Chain Reaction (PCR), Nested PCR, Quantitative Real-Time PCR, Next Generation Sequencing, Loop-Mediated Isothermal Amplification and others are being employed in addition to traditional diagnostic procedures such as ILTV isolation, identification and serological detection. Conventional vaccinations, such as modified live attenuated ILTV vaccines and advanced recombinant vector vaccines expressing diverse ILTV glycoproteins, are used, however these candidates often fail to minimise challenge viral shedding. Certain herbal compounds have been shown to help lessen the effects of disease progression. Additional recombinant and gene-deficient vaccine alternatives are also being explored. Enhanced biosecurity and management approaches, on the other hand, are crucial for better ILT control in ILT-endemic areas.
KEYWORDS: Poultry, Herpes Virus, Chicken, ILT, Infectious Laryngotracheitis, Diagnosis, Control, Review
INTRODUCTION:
Poultry production has been one of the fastest-growing enterprises, with a significant impact on world food security and nutrition. Mondialisation, climate change, and an ever-increasing poultry population have resulted in the emergence of many novel infections in poultry. Infectious laryngotracheitis (ILT), caused by the Gallid herpesvirus1, is one of the poultry respiratory diseases that is progressively spreading around the globe, including the Indian subcontinent that appears in the List B of the Office International des Epizooties (OIE). The virus belongs to the Iltovirus genus, Alphaherpesvirinae subfamily, and Herpesviridae family (Davison, 2010). It is one of the emerging and re - emerging poultry infections (Menendez et al., 2014). Since its first discovery in the world in 1925 (May and Tittsler, 1925), the condition has gained pace throughout centuries, and it has manifested itself as emerging or re-emerging in various regions of the globe in recent years. ILT occurs all over the planet, with the intensity of clinical symptoms and fatality rates ranging from 0% to 70% depending on the severity of the pathogenic strains (Oldoni et al., 2009). The infectious laryngotracheitis virus (ILTV) is a respiratory virus that mostly infects chickens. Pheasants and peafowl are also susceptible to ILTV (Crawshaw and Boycott, 1982).
ECONOMIC SIGNIFICANCE:
Due to significant morbidity and mortality in its acute form, decreased egg production, growth retardation and expenditures devoted on immunization and biosecurity precautions, as well as therapies to combat subsequent infection by other poultry pathogens, ILT can cause colossal economic loss to the poultry sector (Guy and Bagust 2003; Jones 2010; Guy and Garcia 2008; Garcia et al., 2017).
PUBLIC HEALTH SIGNIFICANCE:
There is no evidence that ILTV is communicable to humans or other mammals.
HISTORY:
Infectious laryngotracheitis was first reported in 1925 in United States, and the outbreak that provided the basis for that report occurred in Rhode Island Flock, during October and November 1923. (May and Tittsler, 1925) and then in Australia, the United Kingdom, and Europe (Cover, 1996). Laryngotracheitis, on the other hand, was thought to have existed as early as the 1920s (Cover, 1996). Nasal discharge, edema around the eyes, difficulty in breathing, and the discharge of red bloody mucus were all symptoms of this condition.
Infectious bronchitis, infectious tracheitis, tracheo-laryngitis, laryngotracheitis, chicken "flu," avian diphtheria and Canadian "flu" are some of the old names for ILT (Beach, 1930) and the name infectious laryngotracheitis was given in 1931, by the Special Committee on Poultry Diseases of the American Veterinary Medical Association (Garcia et al., 2017). Beaudette was the first to demonstrate that ILT was caused by a filterable agent (= virus) (Beaudette, 1937). ILT was the first poultry viral disease for which an effective vaccine was developed (Garcia et al., 2017). ILTV could be replicated in the chorioallantoic membrane (CAM) of 10-day-old chicken embryos, according to a report published in 1934 (Burnet, 1934). The virus was identified as a herpesvirus (Cruickshank et al., 1963).
ETIOLOGY:
ILTV belongs to the family Herpesviridae, subfamily Alphaherpesvirinae and is a member of the genus Iltovirus (McGeoch et al., 2000; McGeoch et al., 2006). The virus belongs to Gallid herpesvirus 1 species (Davison et al., 2005).
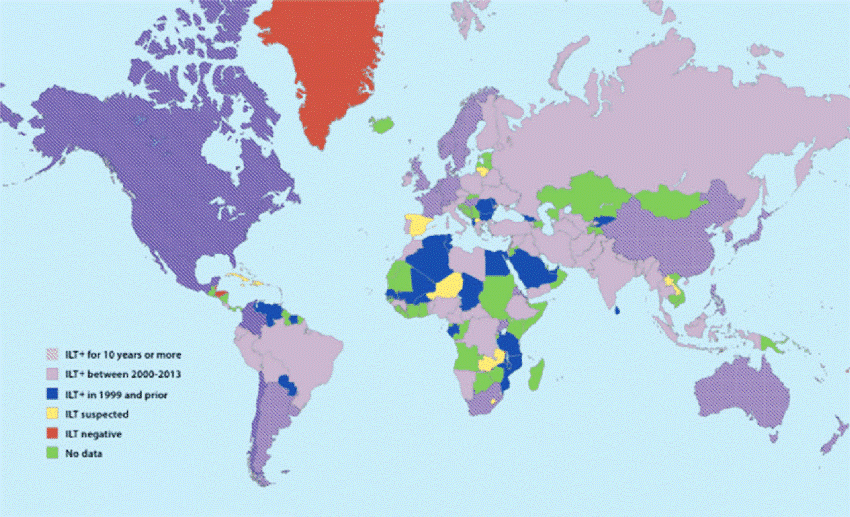
Global distribution of ILT as of 2013 (Menendez et.al., 2014)
CLASSIFICATION:
The International Committee on Taxonomy of Viruses (ICTV) has given a classification of herpesviruses ( www.ictvonline.org ) (Davison, 2010; Davison et al., 2009).
Herpesviruses belong to the order Herpesvirales, which contain three families, three subfamilies, seventeen genera and ninety species (Davison, 2010). Herpesviridae, Alloherpesviridae and Malacoherpesviridae are the three families within the Herpesvirales. The Herpesviridae contain viruses of mammals, birds and reptiles. Alloherpesviridae contain viruses of fish and frog, and Malacoherpesviridae contain viruses of bivalves (Davison et al., 2009). Alphaherpesvirinae, Betaherpesvirinae and gammaherpesvirinae are three subfamilies of the Herpesviridae family.
Alphaherpesvirinae:
Alphaherpesvirinae is an extensive subfamily that contains numerous mammalian and avian viruses (Thiry et al., 2005). Alphaherpesviruses have a short life cycle of 8 to 12 hours, grow rapidly, lyse infected cells, and are capable to establish latency primarily in sensory ganglia (Davison et al., 2009; Fuchs et al., 2000; Thiry et al., 2005). Some alphaherpesviruses are found to have a broad host range (Davison et al., 2009). The Alphaherpesvirinae contains four genera, namely Simplexvirus, Varicellovirus, Mardivirus and Iltovirus (Roizman and Baines, 1991). Herpes simplex virus 1 (HSV1), Herpes simplex virus 2 (HSV2) and Bovine Herpes virus 2 (BHV2) are species under the genus Simplexvirus. The best-known species in the genus Varicellovirus are Swine herpesvirus 1 (SHV1), Bovine herpesvirus 1 (BHV1), Bovine herpesvirus 2 (BHV2), Equine herpesvirus 1 (EHV1), Equine herpesvirus 4 (EHV4), Canine herpesvirus 1 (CHV1) and Feline herpesvirus 1 (FHV1) (Davison, 2010; Davison et al., 2009). Gallid herpesvirus type 2 and 3 (GaHV2 and 3), Anatid herpesvirus 1, Columbid herpesvirus 1 and Meleagrid herpesvirus 1 are avian alphaherpesviruses under genus Mardivirus (Gailbreath and Oaks, 2008; Gennart et al., 2015; Guo et al., 2009; Hunt and Dunn, 2015; Mahony et al., 2015; Petherbridge et al., 2009; Wozniakowski et al., 2013). Gallid herpesvirus type 1 (GaHV1) and Psittacid herpesvirus 1 (PsHV1) are another group of avian Alphaherpesviruses that belong to the genus Iltovirus (Fuchs et al., 2007; Horner et al., 1992; Luppi et al., 2016).
STRUCTURE:
The particles of ILTV have icosahedral symmetry and are 200-350 nm in diameter. The spherical virion comprises core, capsid, tegument, and envelope. The viral genome is a single, linear, double-stranded DNA molecule packaged into a capsid protein. The nucleocapsid of ILTV contains 162 capsomers, which contain 150 hexons and 12 pentons, and the triangulation number (T) is 16. There are 960 copies of the capsid. The diameter of the virion is about 195-250 nm. The envelope surrounding the nucleocapsid is a lipid bilayer, which is associated with the outer surface of the tegument. It contains integral viral glycoproteins (Davison et al., 2005). The genome of ILTV is a linear 155 kb double-stranded DNA, which consists of long and short unique regions (UL, US) and two inverted repeat sequences (internal repeat, IRs; terminal repeat, TRs) that flank the US regions. The genome forms two isomers, which are different from the orientation of the US regions (Davis et al., 1973; Leib et al., 1987). This genome structure is designated as type D herpesvirus genomes (Roizman et al., 2001).

Structure of an ILTV virion.
- Transmission electron microscopy image of an ILTV particle [adapted from Portz et al. (2008)].
- Schematic structure of a virion.
PHYSICO-CHEMICAL PROPERTIES OF ILTV:
ILTV is an enveloped virus and sensitive to ether, chloroform, and other lipolytic solvents (Meulemans et al., 1978). ILTV can be inactivated by ether after 24 hours (Fitzgerald et al., 1963). At 55°C for 10-15 minutes or 38°C for 48 hours, ILTV was readily inactivated (Schalm et al., 1935). However, different strains of ILTV have different resistance to heat. Meulemans et al. in 1978 reported that the Belgian strain had partial infectivity at 56°C for 1 hour. In chicken tracheae and CAMs, ILTV was destroyed in 44 hours at 37°C, or inactivated in 5 hours at 25°C (Cover et al., 1958). It has been shown that the virus was destroyed in 1 min by treating it with 3% cresol or a 1% lye solution (Meulemans et al., 1978). On a chicken farm, 5% hydrogen peroxide mist administered with fumigation equipment completely inactivated ILTV (Neighbour, 1994). At lower temperatures, ILTV maintains infectivity for a long period. The virus survived for 10-100 days in tracheal exudates and chicken carcasses at 13-23°C (Jordan, 1966). When stored at -20°C to -60°C, ILTV was viable for months to years. Storage media containing glycerol or sterile skim milk greatly increases the infectivity in tracheal swabs (Bagust et al., 2000; Schalm et al., 1935).
CHARACTERISTICS OF THE ILTV GENOME:
The genome of ILTV consists of double-stranded linear DNA. Thuree and Keeler in 2006 reported that when 14 different published ILTV genome sequence data from different laboratories were analyzed, ILTV contains 148,665 base pairs, and that the G + C content was 48.16%. It was predicted that the genome had 77 open reading frames (ORFs). Of these ORFs, 63 were homologous to herpes simplex virus-1 genes (McGeoch et al., 2000; Thuree and Keeler, 2006).
VIRUS REPLICATION:
Replication of ILTV is similar to other alpha herpesviruses, such as pseudorabies virus and herpes simplex virus (Prideaux et al., 1992). The virus attaches to receptors on the cell surface. The envelope fuses with the cell membrane and virus nucleocapsid are released into the cytoplasm. Viral DNA is then transported into the nucleus. Transcription of the viral genome, replication of viral DNA, and assembly of new virions occurs in the nucleus (Roizman and Knipe, 2001).
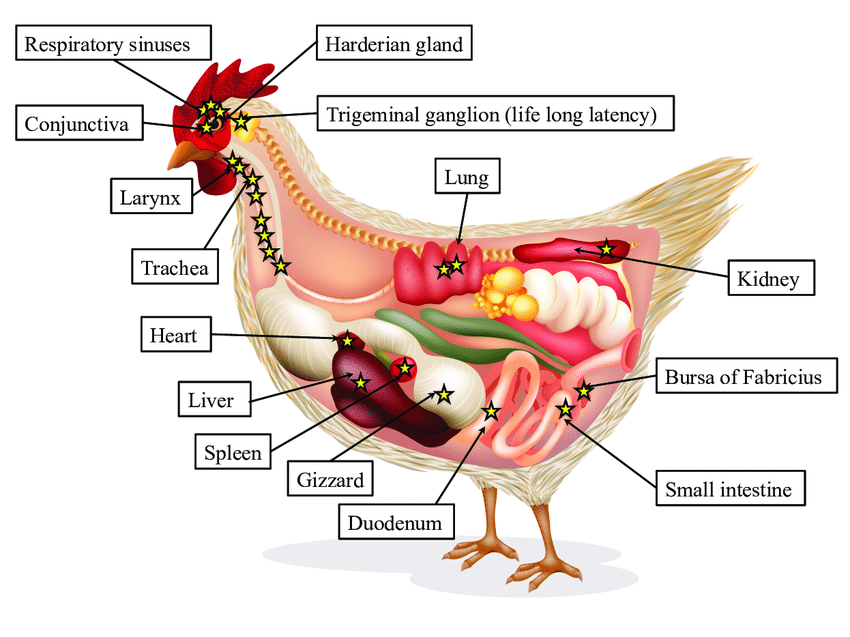
ILTV Pathogenesis.
Stars represent infectious virus. Star number and infectivity are directly proportional to each other. ILTV replicates in the epithelium of larynx, trachea, conjunctiva, respiratory sinuses, air sacs and lungs. Recently, ILTV was observed in kidneys, spleen, liver, heart, gizzard, small and large intestines, bursa of Fabricius and Harderian gland. ILTV establishes a lifelong latency in trigeminal ganglion.
(Adapted from Reddy, A. P., & Reddy, V. (2016))
HOSTS:
All ages of chickens are affected, but chickens older than 3 weeks are most susceptible to ILTV (Fahey et al., 1983). It has been shown that ILTV can infect pheasants, pheasant-bantam crosses, and peafowl (Crawshaw et al., 1982). Subclinical infections with seroconversion were also reported in ducks (Yamada et al., 1980).
Turkeys can be experimentally infected with ILTV; however, turkeys might have an age-dependent resistance, since lesions are only seen in younger birds (Winterfield et al., 1968). Pottz et al. in 2008 reported a natural infection of ILTV in turkeys. Clinical signs in turkeys were similar to chickens (Portz et al., 2008). Starlings, sparrows, crows, pigeons, ducks, and guinea fowls were resistant to ILTV infection (Crawshaw et al., 1982; Hayles et al., 1976; Seddon et al., 1936). Chicken and turkey embryonating eggs can readily propagate ILTV, but guinea fowl and pigeon eggs could not (Jorden, 1966).
Embryonating chicken eggs and several avian cell cultures can be used to propagate ILTV. In chicken embryos, ILTV forms plaques on the chorioallantoic membrane (CAM). The plaques can be observed 48 hours after infection, and embryos might die in 2-12 days post infection. Strains of ILTV showed different plaque size and morphology on the CAM (Burnet et al., 1934; Brandly, 1937; Srinivasan et al., 1977; Hughes et al., 1988).
The ILTV can be propagated in primary cell cultures, such as chicken embryo liver (CEL), chicken embryo kidney (CEK), and chicken kidney (CK) cell cultures (Chang et al., 1973; Hughes and Jones, 1988). Chicken embryo fibroblasts (CEF), Vero cells, and quail cells were not satisfactory for primary isolation of ILTV. Lymphocytes, thymocytes, and activated T cells were not sensitive to ILTV infection (Hughes and Jones, 1988; Schnitzlein et al., 1994).
Viral cytopathic effect (CPE) can be observed in cell culture 4-6 hours PI. The CPE contains swelling of cells, chromatin displacement, and rounding of the nucleoli. The characteristic CPE is syncytia, which forms multinucleated giant cells. Intranuclear inclusion bodies could be observed at 12 hours PI. (Reynolds et al., 1968; Guy and Garcia, 2008).
TRANSMISSION:
Natural transmission of ILTV is through the upper respiratory and ocular routes. Sources of ILTV are clinically affected chickens, latent infected carrier chickens, contaminated dust, litter, and fomites. Egg transmission of the virus has not been verified (Bagust et al., 2000; Guy and Garcia, 2008). Other possible sources of transmission included dog, crows, and cats (Kingsbury et al., 1958). One study showed that wind-borne transmission was critical for ILTV spread (Johnson et al., 2005). After infection, ILTV replicates in the epithelium of the larynx and trachea. Viral particles are present in tracheal tissues and are secreted for 6-8 days PI. The virus may remain in the trachea at 10 days PI (Bagust et al., 1986; Hitchner et al., 1977; Williams et al., 1992).
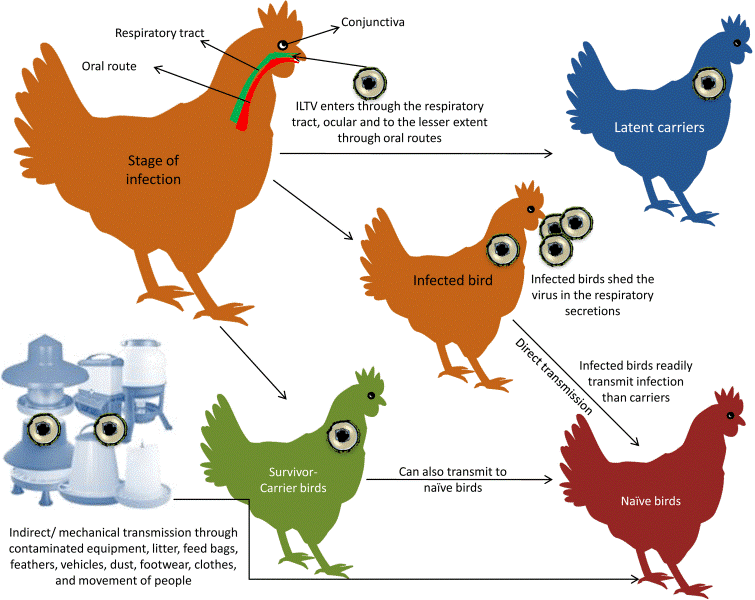
Transmission pattern of ILT virus (Vasudevan Gowthaman et al. 2020)
LATENT INFECTION OF ILTV:
The ILTV can establish latent infections. The virus can be re-isolated from tracheal swabs 7 weeks PI, or two months PI in tracheal samples (Bagust, 1986; Adair et al., 1985). The trigeminal ganglion is the target for latency of ILTV. Four to seven days after ILTV infection by intratracheal route (IT), 40% of infected chickens showed that the virus migrated to trigeminal ganglion, which is nerves system for sensory in face (Bagust, 1986). Fifteen months after vaccination, the latent ILTV in the trigeminal ganglion was reactivated. In mature laying chickens challenged with virulent ILTV, DNA was detected in the trigeminal ganglion by PCR at 31-, 46-, and 61-days PI (Williams et al., 1992). When birds were stressed, such as the onset of lay or re-housing, ILTV can re-activate and spread to susceptible birds (Hughes et al., 1989).
CLINICAL SIGNS:
Clinical signs can be observed 6-12 days PI. Experimental challenge with IT results in a 2–4 day incubation period (Kernohan, 1931; Jordan, 1963). There are two clinical forms of ILT infection (severe and mild). Clinical signs of the severe form include dyspnea and bloody mucus. This form can cause 90%-100% morbidity with mortality ranging from 5% to 70% and average mortality being 10-20% (Bagust et al., 2000; Guy and Garcia, 2008).
The most characteristic signs of the disease can be observed as early as three weeks of age (Dufour-Zavala, 2008). Disease signs generally appear 6-14 days following natural exposure (Jordan, 1966). Infected birds show dyspnea with expectoration of blood-stained mucus and moderate to severe conjunctivitis, respiratory rales, watery eyes, swelling of infraorbital sinuses, mucoid tracheitis and persistent nasal discharge. Furthermore, ILT leads to decreased egg production and reduced weight gain (Garcia et al., 2013), which in turn results in significant economic losses in the poultry industry worldwide (Giambrone, 2008). Besides suffering of the animals, this is the main reason why ILT is a major threat for the poultry industry.
Clinical signs of the mild form include depression, reduced egg production and weight gain, conjunctivitis, swelling of the infraorbital sinuses (almond shaped eyes), and nasal discharge. Morbidity for the mild form is about 5% and mortality 0.1-2%. Generally, it takes 10 to 14 days for recovery, but with some strains the clinical signs may extend for 1-4 weeks (Hinshaw et al., 1931).
GROSS AND MICROSCOPIC LESIONS:
Gross lesions are observed in the larynx and trachea. With the severe form, the mucosa of the respiratory tract shows inflammation and necrosis with haemorrhage. A characteristic feature is intranuclear inclusion bodies in epithelial cells, which are observed about 3 days PI. These cells have a condensed nucleus surrounded by a halo and margining of chromatin. Inclusion bodies are generally present for a few days at the early stage of infection before epithelial cells die. Epithelial cell hyperplasia induces multinucleated cells (syncytia), lymphocytes, histiocytes, and plasma cells, which migrate to the lamina propria. Lesions are followed by desquamation of necrotic epithelium and loss of mucous glands. At this time, bloody mucus in the trachea is observed (Nair and Gough, 2008; Guy and Garcia, 2008). Recently, a solitary case of severe erosive esophagitis and pharyngitis accompanied with epithelial degeneration, necrosis, and syncytia formation with intranuclear inclusion bodies has been reported as an atypical ILT (Sary et al. 2017).
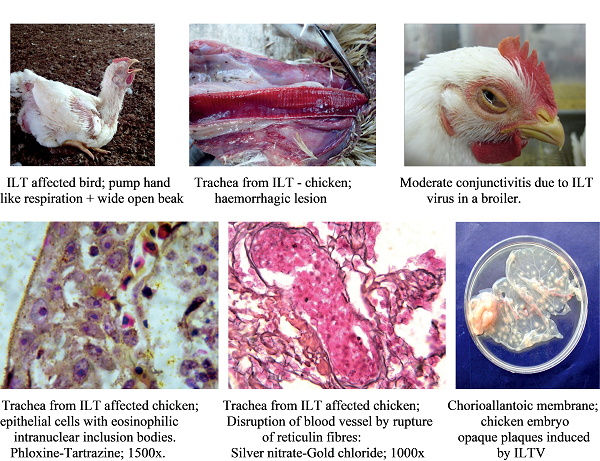
Clinical signs and lesions of ILTV (Sivaseelan, S et al. (2014)
THE DIAGNOSIS AND IDENTIFICATION OF ILTV:
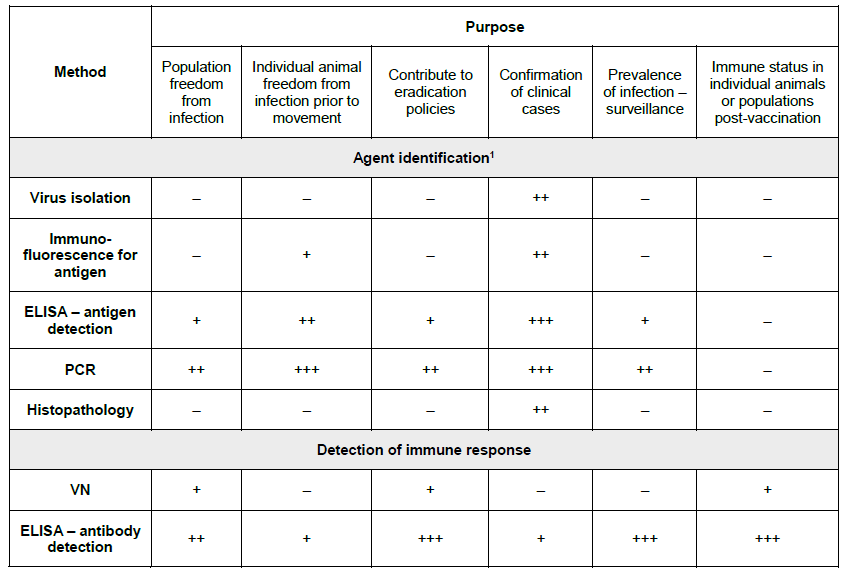
Key: +++ = recommended method; ++ = suitable method; + = may be used in some situations, but cost, reliability, or other factors severely limits its application; – = not appropriate for this purpose. Although not all of the tests listed as category +++ or ++ have undergone formal validation, their routine nature and the fact that they have been used widely without dubious results, makes them acceptable. ELISA = Enzyme-linked immunosorbent assay; PCR = polymerase chain reaction; VN = virus neutralisation. (OIE Terrestrial Manual, 2018)
Laboratory diagnosis is required for ILT, because other diseases cause similar clinical signs and lesions, such as infectious bronchitis (IB), Newcastle disease (ND), avian influenza (AI), infectious coryza, and Mycoplasmosis. Standard procedures for ILTV diagnosis include clinical signs, gross and microscopic lesions, and virus isolation. The histopathologic lesions associated with ILTV infection include intranuclear inclusion bodies in tracheal epithelial cells (Nair et al., 2008; Guy et al., 2008). ILTV infection can be confirmed using several methods, including virus isolation, DNA detection, and serologic tests. For ILTV isolation, the CAM inoculation of 9-to-12-day-old embryos and primary cell culture are used. Samples from the trachea, conjunctiva, larynx, and lung of clinically affected birds are collected and inoculated on the CAM (Hughes et al., 1988). Plaques can be observed 2 days PI; however, they usually develop on the CAM 5-7 days PI. Plaque size and morphology on the CAM can be used to differentiate the virulence of strains. The CEL and CK cell cultures are suitable for ILTV isolation. Multinucleated giant cells may be observed 24 hours PI (Guy and Garcia 2008).
The immunoperoxidase (IP) and FA tests with labeled monoclonal antibodies are used to detect ILTV in tracheal smears (Guy et al., 1992). The AC-ELISA and ELISA can be used for ILTV antigen and antibody detection (York et al., 1988; Chang et al., 2002). ILTV can be isolated in embryonic eggs inoculated via the CAM route. Primary chicken cells, such as chicken embryo liver (CEL), chicken embryo kidney (CEK), and chicken kidney (CK) are also used for ILTV isolation (Srinivasan et al., 1977; Hughes et al., 1988).
Traditional antigen detection uses ILTV polyclonal or monoclonal antibodies to bind ILTV antigen from clinical samples. Viral antigen was detected using direct or indirect fluorescent antibodies (FA) in the tracheal smear or tracheal tissues (Goodwin et al., 1991). A more sensitive method using immunoperoxidase (IP) labelled monoclonal antibodies can be used as immunoprobes to detect ILTV in tracheal smears. This IP method detected ILTV on the second day PI (Guy et al., 1992). Agar gel immunodiffusion (AGID) uses hyperimmune serum against ILTV to detect antigen in tracheal samples and it can differentiate ILT from the diphtheritic form of fowl pox. However, the sensitivity was lower than other methods (Jorden et al., 1962). Antigen capture enzyme-linked immunosorbent assay (AC-ELISA) uses ILTV monoclonal antibodies for antigen detection. The AC-ELISA was faster and more accurate than AGIP or FA (York and Fahey, 1988).
ILT DNA detection methods have developed rapidly in recent years. These methods can identify ILTV quickly, accurately, and are highly sensitive. Molecular techniques for ILTV detection include cloned DNA probes for dot-blot hybridization (Nagy, 1992), PCR (Abbas et al., 1996; Chang et al., 1997; Clavijo et al., 1997; Alexander et al., 1998), nested PCR (Humberd et al., 2002), real-time PCR (Creelan et al., 2006), multiplex PCR (Pang et al., 2002), in situ hybridization (Nielsen et al., 1998), and PCR followed by restriction fragment length polymorphism (RFLP) (Chang et al., 1997; Kirkpatrick et al., 2006; Oldoni and Garcia, 2007; Oldoni et al., 2008). Comparing the conventional methods of ILTV detection with PCR has reported that PCR was more sensitive than virus isolation in cell culture and electron microscopy. PCR also detected ILTV in the samples, which was contaminated with other pathogens (Williams et al., 1994).
ILTV STRAIN DIFFERENTIATION:
It is difficult to identify different strains of ILTV by serological methods, because the ILTVs have close immunodominant domains (Shibley et al., 1962). The most common and effective molecular method for ILTV differentiation is PCR followed by restriction fragment length polymorphism (RFLP). PCR-RFLP analysis of single or multiple viral genome regions can differentiate strains from various geographic areas and vaccine from wild (field) strains. (Leib et al., 1986; Keeler et al., 1993; Chang et al., 1997; Oldoni et al., 2007 and 2008; Neff et al., 2008; Oldoni et al., 2009). Restriction endonuclease analysis of ILTV DNA can differentiate vaccine strains from wild type strains (Guy et al., 1989).
Researchers demonstrated that multiple gene PCR-RFLP was more reliable to differentiate vaccines from field strains (Kirkpatrick et al., 2006).
Oldoni and Garcia (2007) investigated ILTV isolates from commercial poultry that were collected between 1988 and 2005 using multiple gene PCR-RFLP analysis (ORFB-TK, ICP4, UL47/gG, and gM/ UL9). They were able to separate ILTVs into nine genetic groups. Group I and II comprised the USDA reference strain and tissue culture origin (TCO) vaccine strains. Group IV isolates were identical to chicken egg origin (CEO) vaccine strains, whereas group V isolates, which had one PCR-RFLP pattern different from the CEO vaccine strains are CEO-related isolates. Group III, VI, VII, VIII, and IX were field ILTV strains with genomic types different from CEO and TCO vaccines. In that report, most of ILTV positive poultry isolates were related to vaccine strains (Oldoni & Garica, 2007). Oldoni et al. in 2008 investigated 46 ILTV field isolates collected in the U.S. from 2006 to 2007. After multiple gene PCR-RFLP analysis, most isolates (63%) were closely related to vaccine strains (group III, IV, and V) (Oldoni et al., 2008). According to these reports, most ILTV field isolates in the U.S. were derived from vaccines.
DIFFERENTIAL DIAGNOSIS:
The other respiratory diseases exhibiting similar clinical disease must be differentiated from ILT. The
diphtheritic lesions induced by ILT spread over the whole length of trachea and resemble lesions induced by the fowlpox virus (Tripathy and Reed 2013). Tracheal lesions in mild or low virulent form of ILTV is similar to that of lesions caused by other respiratory pathogens such as avian influenza virus, Newcastle disease virus, infectious bronchitis virus and fowl adenovirus (Davidson et al. 2015).
TREATMENT:
No drug has been shown to be effective in reducing the severity of lesions or relieving disease signs. If a diagnosis of LT is obtained early in an outbreak, vaccination of unaffected birds may induce adequate protection before they become exposed.
IMMUNOLOGY AND VACCINATION:
ILT vaccination induces partial protection in one week against challenge at 3-4 days post infection (PI). Humoral immunity is not the major immune response against ILTV in chickens. Research verified the importance of cell-mediated immunity (CMI) in the infection of ILTV resistance. Mucosal antibodies were not essential for resistance to challenge (Fahey et al., 1990).
Vaccination is effective to prevent ILTV infection. However, ILT vaccine viruses can create latent infected carrier chickens. These latent carriers are a source for spread of virus to non-vaccinated flocks. Therefore, it is recommended that ILT vaccines be used only in areas where ILT is endemic. The most currently used ILT vaccine strains are attenuated modified-live chicken-embryo-origin (CEO) or tissue-culture-origin (TCO) viruses. Compared with protection afforded by TCO and CEO vaccines, there was no significant difference in the immunity of chickens at 10 weeks PI. However, when chickens over 20 weeks of age were vaccinated, the CEO vaccines induced better protection than TCO vaccines (Andreason et al., 1989).
Methods for vaccine administration are eye drop, drinking water, and aerosol spray. The drinking water route poses some problems in that chickens might not receive enough virus at the target organ (nasal epithelial cells) and drinking water quality varies between poultry houses. Thus, these birds may fail to develop protective immunity and may have rolling (continual) reactions (Robertson et al., 1981). On the other hand, with spray route, some chickens may develop severe reactions, because excess dosages of small droplets can penetrate deep into the respiratory tract (Clark et al., 1980).
There are several disadvantages when using modified-live ILT vaccines. They include: insufficient attenuation, latent infected carriers, and the spreading of virus to non-vaccinated flocks. Reports have shown that modified-live vaccines increase their virulence by bird-to-bird passage (Guy et al., 1991; Kotiw et al., 1995). CEO vaccines have the tendency to increase in virulence more than TCO vaccines, when passed in chickens (Guy et al., 1991).
Investigations of ILTV isolates collected from around the world were analyzed by PCR-RFLP. They revealed that current wild virulent isolates were closely related to vaccine strains. This implies that field isolates originated from vaccine strains after back passage in chickens (Leib et al., 1986; Keeler et al., 1993; Chang et al., 1997; Oldoni et al., 2008; Neff et al., 2008; Oldoni et al., 2009).
Recent molecular advances have developed recombinant vaccines. Researchers have developed other vaccines using inserted partial ILTV genes into fowlpox and HVT modified genomes. A recombinant fowlpox vaccine, which contained ILTV gB gene, was shown to induce protection against virulent strains and the protection was similar to attenuated-live vaccines (Tong et al., 2001). Another recombinant fowlpox virus, which contains ILTV gB and UL 32 genes, was shown to provide protection against virulent strain challenge (Davison et al., 2006). Two licensed commercial recombinant ILT vaccines are used around the world. One is produced by the CEVA (Biomune Company, Lenexa KS), which uses fowl poxvirus as a vector with an insertion of ILTV gene. The other is produced by Intervet (Intervet Inc. Millsboro, DE), in which ILTV genes are cloned into a herpesvirus of turkey (HVT). They have shown some efficacy in the field.
Several studies tried to develop new ILT vaccine candidates by gene deletion. Some ILTVs, with deleted virulent viral genes, retained their ability to induce immune responses without producing clinical signs. Recombinant virus with deleted gJ, TK, and, UL0 genes readily showed attenuation, and could be used for vaccine production (Okamura et al., 1994; Schnitlzlein et al., 1995; Veits et al., 2003b). The gG-deficient ILTV administered by either eye-drop or drinking- water routes induce adequate immunity against challenge. Therefore, it may have a use for large-scale vaccination (Devlin et al., 2008). There were also ILTV non-essential genes, which were deleted to test their ability as vaccines. The ILT mutants, such as deleted five unique open reading frames (ORF A-E), removed gN and gM, and the green fluorescent protein was inserted into the UL50 gene deleted region, could be used as recombinant ILT vaccines (Fuchs et al., 1999; Fuchs et al., 2005; Fuchs et al., 2000). ILT recombinant vaccines used ILTV as a viral vector to contain foreign viral genes. One ILTV vaccine contains H5 and H7 genes of highly pathogenic AIVs. This recombinant ILTV may be used as bivalent vaccine against ILT and pathogenic AIV (Veits et al., 2003b).
PREVENTION OF ILTV INFECTION USING CHICKEN HOUSE MANAGEMENT:
Field isolates and vaccine viruses can establish latent infected carriers. Thus, it is important to avoid contact between vaccinated or recovered field virus infected birds with non-vaccinated chickens. It is also critical to remove contaminated fomites for prevention and control of ILTV infection. To control ILTV outbreaks, improved biosecurity and management practices are necessary. Biosecurity includes protocols and procedures to prevent pathogens from infecting and transmitting disease by humans, insects, wild birds, or other animals (Kingsbury et al., 1958).
CONCLUSION & RECOMMENDATIONS:
ILT continues to pose a serious danger to the global poultry sector. Enhanced understanding on virus biology, epidemiology, and pathogenesis, as well as rigorous biosecurity, may aid in the control of disease epidemics. Rapid diagnosis, strong biosecurity, a vaccination regimen, the use of GIS technology, and adequate washing, disinfection, and heating of poultry are all part of the integrated approach. The most successful technique in reducing ILTV will be a coordinated plan that includes early diagnosis, tight biosecurity, immunisation, GIS technology, thorough cleaning, disinfection, and heating of chicken houses, and greater communication between government and industry.
CITATIONS:
Abbas, F., Andreasen, J. R., Baker, R. J., Mattson, D. E., and Guy, J. S., 1996. Characterization of monoclonal antibodies against infectious laryngotracheitis virus. Avian Dis. 40, 49-55.
Adair, B. M., Todd, D., McKillop, E. R, and Burns, K., 1985. Comparison of serological tests for detection of antibodies to infectious laryngotracheitis virus. Avian Pathol.14, 461-469.
Alexander, H.S., Key, D. W., and Nagy, E., 1998. Analysis of infectious laryngotracheitis virus isolates from Ontario and New Brunswick by polymerase chain reaction. Can. J. Vet. Res. 62, 68-71.
Andreasen, J. R. Jr, Glisson, J. R, Goodwin, M. A, Resurreccion, R. S, Villegas, P., and Brown, J., 1989. Studies of infectious laryngotracheitis vaccines: Immunity in layers. Avian Dis.33, 524-530.
Bagust, T. J., 1986. Laryngotracheitis (Gallid-1) herpesvirus infection in the chicken. 4 Latency establishment by wild and vaccine strains of ILT virus. Avian Pathol. 6(15), 581-595.
Bagust, T.J., Jones, R.C., and Guy, J. S., 2000. Avian infectious laryngotracheitis. Rev. Sci. Tech. Off. Int. Epiz. 19 (2), 483-492.
Beaudette, F. R. (1937). Infectious laryngotracheitis. Poultry Science, 16(2), 103-105.
Beach J. R. (1930). The virus of laryngotracheitis of fowls. Science (New York, N.Y.), 72(1877), 633–634.
Brandly, C. A., 1937. Studies on certain filterable viruses. I. Factors concerned with the egg propagation of fowl pox and infectious laryngotracheitis. J. Am. Vet. Med. Asso. 90, 479-487.
Burnet F. M. (1934). The Propagation of the Virus of Infectious Laryngo-Tracheitis on the Chorio-Allantoic Membrane of the Developing Egg. British Journal of Experimental Pathology, 15(1), 52–55.
Chang, P. W., Jasty, V., Fry, D., and Yates, V. J., 1973. Replication of a cell-culture-modified infectious laryngotracheitis virus in experimentally infected chickens. Avian Dis. 17, 683-689.
Chang, P. C., Lee, Y. L., Shien, J. H., and Shieh, H. K., 1997. Rapid differentiation of vaccine strains and field isolatesof infectious laryngotracheitis virus by restriction fragment length polymorphism of PCR products. J. Virol. Meth. 66, 179-186.
Chang, P. C., Chen, K. T., Shien, J. H., and Shieh, H. K., 2002. Expression of infectious laryngotracheitis virus glycoproteins in Escherichia coli and their application in enzyme-linked immunosorbent assay. Avian Dis. 46, 570-580.
Clarke, J. K., Robertson, G. M., and Purcell, D. A., 1980. Spray vaccination of chickens using infectious laryngotracheitis virus. Aus.Vet. J. 56, 424-428.
Clavijo, A., and Nagy, E., 1997. Differentiation of infectious laryngotracheitis virus strains by polymerase chain reaction. Avian Dis. 41, 241-246.
Cover, M. S., and Benton, W. J., 1958. The biology variation of infectious laryngotracheitis virus. Avian Dis. 2, 375-383.
Cover, M. (1996). The Early History of Infectious Laryngotracheitis. Avian Diseases, 40(3), 494-500.
Crawshaw, G. J., and Boycott, B. R., 1982. Infectious laryngotracheitis in peafowl and pheasants. Avian Dis. 26, 397-401
Creelan, J. L., Calvert, V. M., Graham, D. A., and McCullough, S. J., 2006. Rapid detection and characterization from field cases of infectious laryngotracheitis virus by real-time polymerase chain reaction and restriction fragment length polymorphism. Avian Pathol. 35, 173-179.
Cruickshank, J. G., Berry, D. M., & Hay, B. (1963). The fine structure of infectious laryngotracheitis virus. Virology, 20(2), 376-378.
Davidson I, Raibstein I, Altory A. 2015. Differential diagnosis of fowlpox and infectious laryngotracheitis viruses in chicken diphtheritic manifestations by mono and duplex real-time polymerase chain reaction. Avian Pathol. 44(1): 1–4.
Davison A. J. (2010). Herpesvirus systematics. Veterinary microbiology, 143(1), 52–69.
Davison, A. J., Eberle, R., Hayward, G. S., McGeoch, D. J., Minson, A. C., Pellett, P. E., Roizman, B., Studdert, M. J., and Thiry, E., (2005). Herpesviridae. In: Fauquet, C. M., Mayo, M. A., Maniloff, J., Desselberger, U., and Ball, L. A. (Eds.), Virus Taxonomy: Eighth Report of the International Committee on Taxonomy of Viruses. Elsevier Academic Press, San Diego, pp. 193-212.
Davison, A.J., Eberle, R., Ehlers, B., Hayward, G.S., McGeoch, D.J., Minson, A.C., Pellett, P.E., Roizman, B., Studdert, M.J., Thiry, E., (2009). The order Herpesvirales. Arch Virol 154, 171-177.
Davison, S. E., Gingerich, N., Casavant, S., and Eckroade, R. J., (2006). Evaluation of the efficiency of a live fowlpox-vectored infectious laryngotracheitis/avian encephalomyelitis vaccine against ILT viral challenge. Avian Dis. 50, 50-54.
Devlin, J. M., Browning G. F., Gilkerson, J. R., Fenton, S. P., and Hartley, C. A., (2008). Comparison of the safety and protective efficacy of vaccination with glycoprotein-G-deficient infectious laryngotracheitis virus delivered via eye-drop, drinking water or aerosol. Avian Pathol. 37, 83-88.
Dufour-Zavala L. 2008. Epizootiology of infectious laryngotracheitis and presentation of an industry control program. Avian Dis. 52(1):1–7.
Fahey, K. J, and York, J. J., 1990. The role of mucosal antibody in immunity to infectious laryngotracheitis virus in chickens. J. Gen. Virol. 71, 2401-2405.
Fahey, K. J., Baugst, T. J., and York, J. J., 1983. Laryngotracheitis herpesvirus infection in the chicken: the role of humoral antibody in immunity to a grade challenge infection. Avian Pathol. 12, 505-514.
Fitzgerald, J. E. and Hanson, L. E., 1963. A comparison of some properties of laryngotracheitis and herpes simplex viruses. Am. J.Vet. Res. 24, 1297-1303.
Fuchs, W., and Mettenleiter, T. C., 1999. DNA sequence of the UL6 to UL20 of infectious laryngotracheitis virus and characterization of the UL10 gene product as a nonglycosylated and nonessential virion protein. J. Gen. Virol. 80, 2173-2182.
Fuchs, W., and Mettenleiter, T. C., 2005. The nonessential UL49.5 gene of infectious laryngotracheitis virus encodes an O-glycosylated protein which forms a complex with the non-glycosylated UL10 gene product. Virus Res. 112, 108-114.
Fuchs, W., Veits, J., Helferich, D., Granzow, H., Teifke, J.P., Mettenleiter, T.C., 2007. Molecular biology of avian infectious laryngotracheitis virus. Vet Res 38, 261-279.

